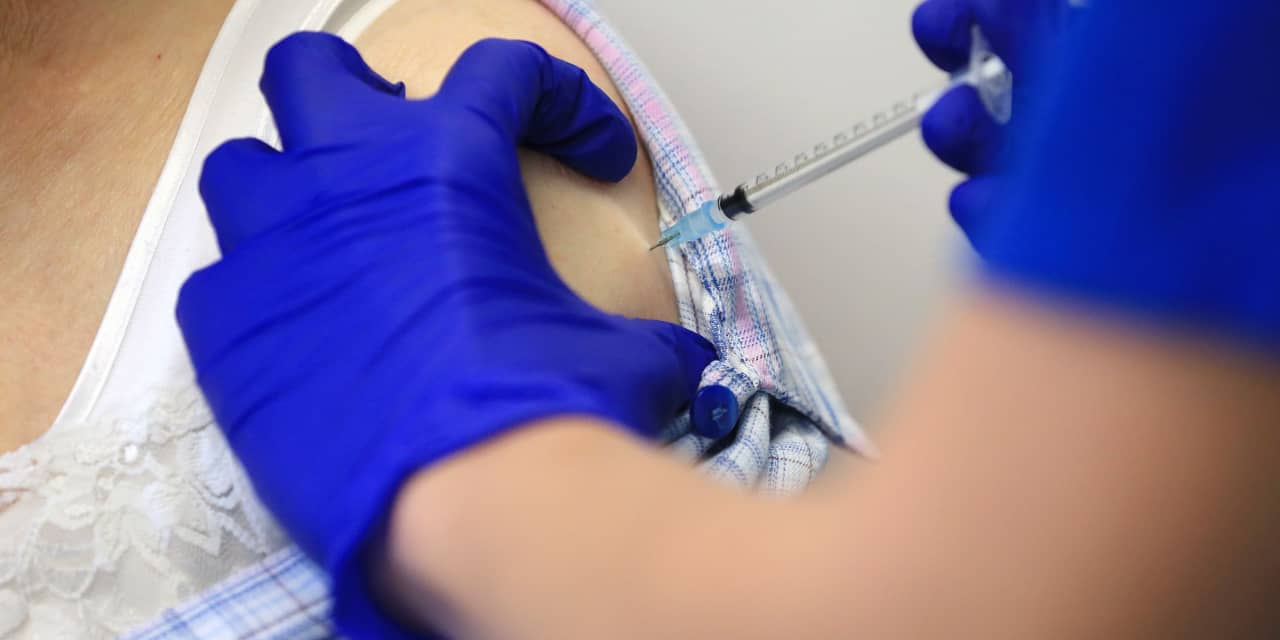
The UK National Health Service has pushed back the window for people to receive a second dose of the COVID-19 vaccine made by Pfizer and Bayonet – companies tested on only a small percentage of patients.
The NHSA wrote a letter to hospitals stating that they were to receive a second dose after January. 4 should be rescheduled to 12 from the three-week period, most recipients booked in the last week of that deadline. The move will bring the window closer to the deadline for the only-approved vaccine developed by Oxford University and the pharmaceutical company Strazeneca AZN.
Read: UK Strazeneca – Oxford COVD-19 vaccine approved, the UK now has enough vaccines to ‘cover the whole population’
The NHSA said the move would provide the greatest protection to those at risk in the short term and have the greatest impact on mortality.
There has been an increase in cases of coronavirus confirmed in the last month, with the UK saying the new form of the virus that causes Covid-19 is more easily transmissible. On November 30, the daily cases increased from 12,330 at the end of November to 50,023.
When the UK’s Medicines and Healthcare Products Regulatory Agency approved its emergency by vaccinating US pharmaceutical company Pfizer PFE,
And its German partner BNTX,
He said the interval between doses should be at least three weeks, and is based on an analysis that included patients who received their second vaccination within 19 to 42 days after the first vaccination.
Pfizer said the safety and efficacy of the vaccine have not been tested in the new interval. “Phase 3 study of Pfizer and bayonet tech for COVID-19 vaccine, 21-day separate, after 2-dose schedule, was designed to evaluate vaccine safety and efficacy. “The safety and efficacy of the vaccine were not evaluated at different dosing schedules because most trial participants received a second dose within the window specified in the study design,” the company said.
Pfizer also noted that there are no data to show that safety is maintained even 21 days after receiving the first dose.
“While decisions about alternative dosing methods are with health officials, Pfizer believes that those serious health officials make efforts to monitor any alternative schedules implemented and give each recipient the maximum possible protection, meaning vaccination with two doses of the vaccine.” . ” Said Pfizer. The message with Biotech was not immediately returned.
U.S. In a briefing document provided by the drug companies to the Food and Drug Administration, they said that about 0% of patients receiving a second dose administered it within 10 weeks of receiving the first drug.
.