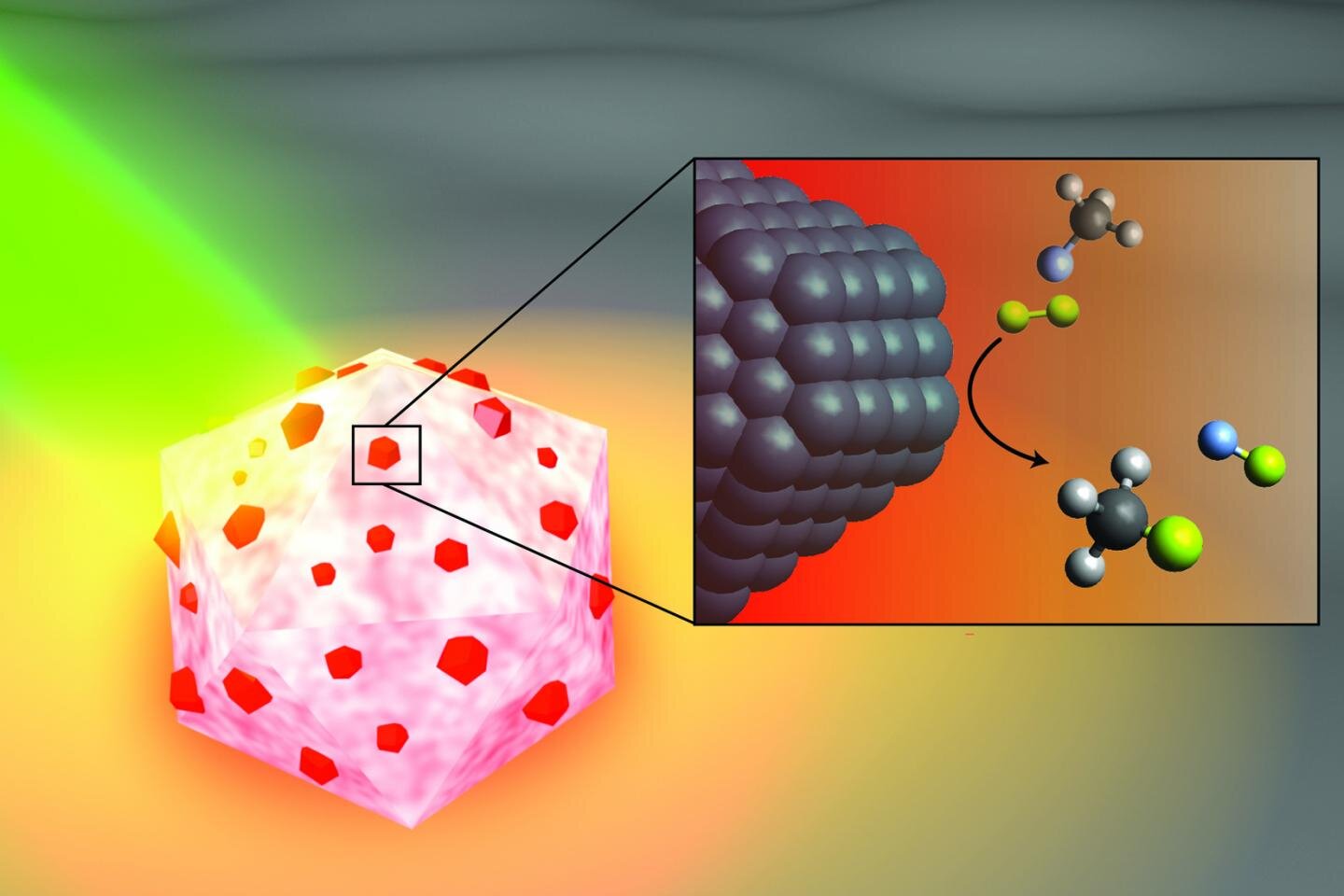

An Artistic Illustration of the Light Activated Antenna-Reactor Catalyst Rice University engineers designed to break down carbon-fluorine bonds in fluorocarbons. The aluminum portion of the particle (white and pink) captures light energy (green), activating islands of palladium catalysts (red). In the box, fluoromethane molecules (above) made up of one carbon atom (black), three hydrogen atoms (gray), and one fluorine atom (light blue) react with the deuterium (yellow) molecules near the surface from palladium (black), cleaving carbon – a fluorine bond to produce deuterium fluoride (right) and monodeuterate methane (bottom). Credit: H. Robatjazi / Rice University
Rice University engineers have created a lightweight catalyst that can break the strong chemical bonds in fluorocarbons, a group of synthetic materials that include persistent environmental pollutants.
In a study published this month in Catalysis of nature, Naomi Halas, pioneer in nanophotonics at Rice, and collaborators from the University of California, Santa Barbara (UCSB) and Princeton University demonstrated that small aluminum spheres dotted with palladium speckles could break carbon-fluorine (CF) bonds to through a catalytic process known as hydrodefluorination in which a fluorine atom is replaced by a hydrogen atom.
The strength and stability of CF bonds are behind some of the most recognized chemical brands of the 20th century, such as Teflon, Freon, and Scotchgard. But the strength of those bonds can be problematic when fluorocarbons enter the air, soil, and water. Chlorofluorocarbons, or CFCs, for example, were banned by an international treaty in the 1980s after they were discovered to destroy Earth’s protective ozone layer, and other fluorocarbons were on the “chemicals forever” list. selected by a 2001 treaty.
“The hardest part of remedying any of the fluorine-containing compounds is breaking the CF bond; it takes a lot of energy,” said Halas, an engineer and chemist whose Nanophotonics Laboratory (LANP) specializes in creating and studying nanoparticles that interact with light .
For the past five years, Halas and his colleagues have pioneered methods of making “antenna reactor” catalysts that stimulate or accelerate chemical reactions. While catalysts are widely used in industry, they are generally used in energy intensive processes that require high temperature, high pressure, or both. For example, a mesh of catalytic material is inserted into a high-pressure container in a chemical plant, and natural gas or other fossil fuel is burned to heat the gas or liquid flowing through the mesh. LANP’s antenna reactors dramatically improve energy efficiency by capturing light energy and inserting it directly at the point of the catalytic reaction.
At Catalysis of nature According to the study, the energy capture antenna is a smaller aluminum particle than a living cell, and the reactors are palladium islands scattered across the aluminum surface. The energy saving feature of antenna reactor catalysts is perhaps best illustrated by another of Halas’ previous successes: solar steam. In 2012, her team demonstrated that its energy-collecting particles could instantly vaporize water molecules near its surface, meaning that Halas and her colleagues could produce steam without boiling water. To clarify the point, they showed that they could steam with ice water.
The antenna reactor catalyst design allows the Halas team to mix and match metals that are best suited to capture light and catalyze reactions in a particular context. The work is part of the green chemistry movement towards cleaner and more efficient chemical processes, and LANP has previously demonstrated catalysts to produce ethylene and synthesis gas and to split ammonia to produce hydrogen fuel.
The study’s lead author, Hossein Robatjazi, Beckman Postdoctoral Fellow at UCSB, earned his Ph.D. Rice in 2019 did most of the research during his graduate studies in the Halas lab. He said the project also shows the importance of interdisciplinary collaboration.
“I finished the experiments last year, but our experimental results had some interesting features, changes in reaction kinetics under illumination, that raised an important but interesting question: What role does light play in promoting CF breakdown chemistry? ” he said.
The answers came after Robatjazi came for his postdoctoral experience at UCSB. He was assigned the task of developing a microkinetic model, and a combination of model ideas and theoretical calculations by Princeton’s collaborators helped explain the puzzling results.
“With this model, we use the surface science perspective on traditional catalysis to uniquely link experimental results with changes in the reaction pathway and reactivity under light,” he said.
Demonstration experiments on fluoromethane could be just the beginning for the CF breakdown catalyst.
“This general reaction can be helpful in remedying many other types of fluorinated molecules,” said Halas.
Low-temperature photocatalyst could reduce synthesis gas carbon footprint
Hossein Robatjazi et al, Plasma-Driven Carbon-Fluorine (C (sp3) -F) Binding Activation with Mechanistic Perceptions of Pathways Mediated by Hot Carriers, Catalysis of nature (2020). DOI: 10.1038 / s41929-020-0466-5
Provided by Rice University
Citation: The team presents a catalyst that can break the troublesome CF bonds (2020, June 22) recovered on June 23, 2020 from https://phys.org/news/2020-06-team-unveils-catalyst-problematic- cf.html
This document is subject to copyright. Other than fair dealing for private research or study purposes, no part may be reproduced without written permission. The content is provided for informational purposes only.