[ad_1]

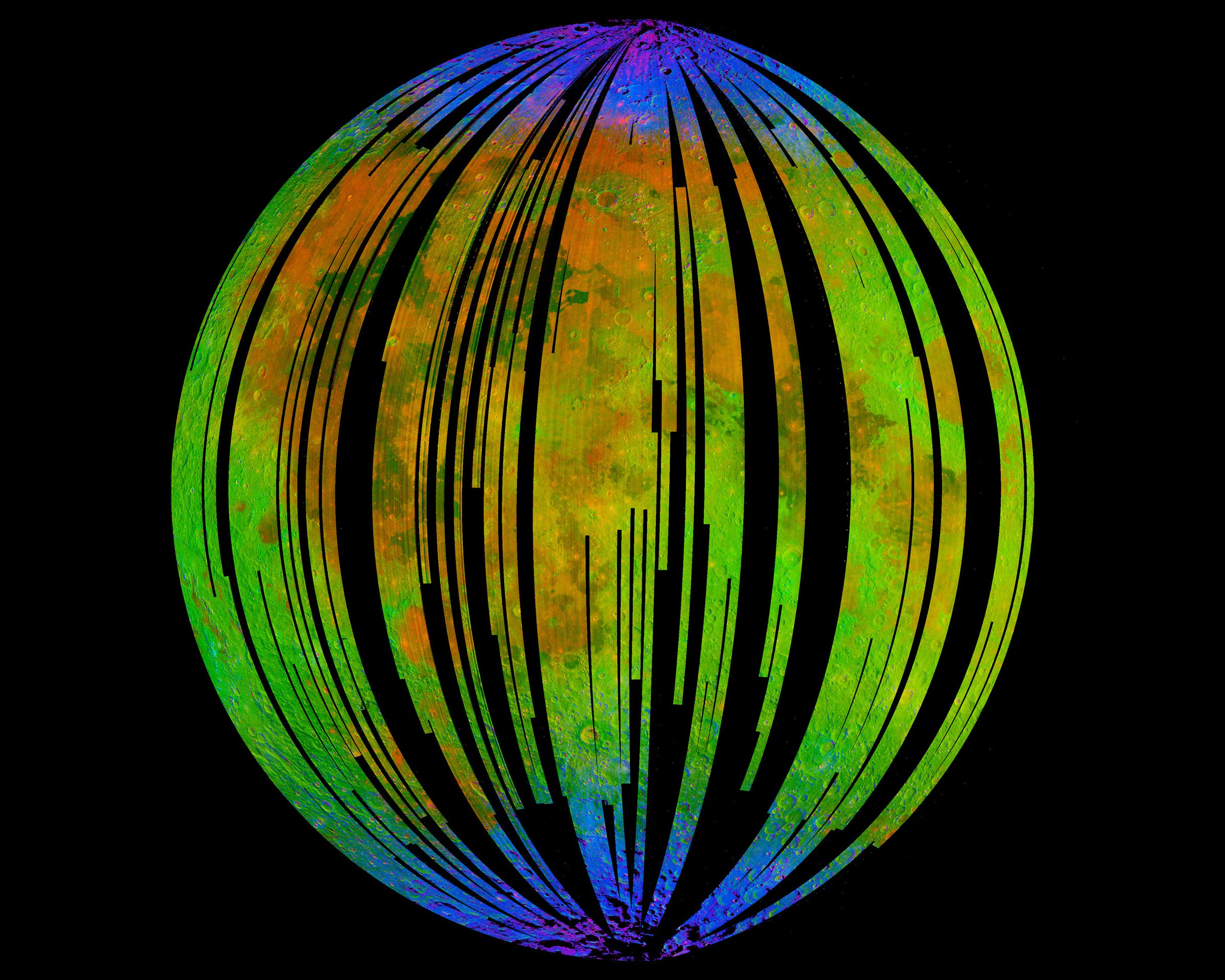
Blue areas in this composite image from the Moon Mineralogy Mapper (M3) aboard the Indian Space Research Organization’s Chandrayaan-1 orbiter show concentrated water at the Moon’s poles. By concentrating on the spectra of the rocks there, the researcher found signs of hematite, a form of rust. Credit: ISRO / NASA / JPL-Caltech / Brown University / USGS
While our Moon is airless, research indicates the presence of hematite, a form of oxide that normally requires oxygen and water. That has scientists baffled.
Mars has long been known for its rust. The iron on its surface, combined with water and oxygen from the ancient past, gives the Red Planet its hue. But scientists were recently surprised to find evidence that our airless Moon also has rust.
A new article in Science Advances reviews data from the Indian Space Research Organization’s Chandrayaan-1 orbiter, which discovered water ice and mapped a variety of minerals while studying the Moon’s surface in 2008. Lead author Shuai Li of the University of Hawaii has studied water extensively in data from the Moon Mineralogy Mapper instrument of Chandrayaan-1, or M3, which was built by POTJet Propulsion Laboratory in Southern California. Water interacts with rock to produce a variety of minerals and M3 detected spectra, or light reflected off surfaces, which revealed that the Moon’s poles had a composition very different from the rest.
Intrigued, Li focused on these polar spectra. While the surface of the Moon is littered with iron-rich rocks, he was surprised to find a close match to the spectral signature of hematite. The mineral is a form of iron oxide, or rust, that is produced when iron is exposed to oxygen and water. But the Moon is not supposed to have oxygen or liquid water, so how can it be rusty?
Metal mystery
The mystery begins with the solar wind, a stream of charged particles that flows from the Sun, bombarding the Earth and Moon with hydrogen. Hydrogen hinders the formation of hematite. It is what is known as a reducer, which means that it adds electrons to the materials with which it interacts. That’s the opposite of what is needed to make hematite: for iron to oxidize, it requires an oxidizer, which removes electrons. And while the Earth has a magnetic field that protects it from this hydrogen, the Moon does not.
“It’s very puzzling,” Li said. “The Moon is a terrible environment for hematite to form.” So she turned to. JPL scientists Abigail Fraeman and Vivian Sun to help push M3data and confirm their discovery of hematite.
“At first, I didn’t quite believe it. It shouldn’t exist based on the conditions present on the Moon, ”Fraeman said. “But ever since we discovered water on the Moon, people have been speculating that there might be a greater variety of minerals than we think if that water had reacted with the rocks.”
After looking closely, Fraeman and Sun were convinced that M3In fact, the data indicate the presence of hematite at the lunar poles. “In the end, the spectra contained hematite convincingly, and there needed to be an explanation for why it’s on the Moon,” Sun said.
Three key ingredients
His article offers a three-point model to explain how rust might form in such an environment. For starters, although the Moon lacks an atmosphere, it does indeed harbor traces of oxygen. The source of that oxygen: our planet. Earth’s magnetic field creeps behind the planet like a windsock. In 2007, the Japanese orbiter Kaguya discovered that oxygen from Earth’s upper atmosphere can travel in this magnetic tail, as it is officially known, traveling the 239,000 miles (385,000 kilometers) to the Moon.
That discovery fits the data from M3, which found more hematite on the near side of the Moon facing Earth than on its far side. “This suggested that oxygen from the Earth might be driving the formation of hematite,” Li said. The Moon has drifted slowly away from Earth for billions of years, so it is also possible that more oxygen passed through this rift when the two were closer together in the ancient past.
Then there is the matter of all that hydrogen being delivered by the solar wind. As a reducing agent, hydrogen should prevent oxidation from occurring. But the Earth’s magnetic tail has a mediating effect. In addition to transporting oxygen to the Moon from our home planet, it also blocks over 99% of the solar wind during certain periods of the Moon’s orbit (specifically, when it is in the full moon phase). That opens occasional windows during the lunar cycle when rust can form.
The third piece of the puzzle is water. While most of the Moon is completely dry, water ice can be found in the shaded lunar craters on the opposite side of the Moon. But the hematite was detected far from that ice. Instead, the article focuses on the water molecules found on the lunar surface. Li proposes that fast-moving dust particles that regularly hit the Moon could release these surface-borne water molecules, mixing them with iron in the lunar soil. The heat from these impacts could increase the rate of oxidation; the dust particles themselves may also be transporting water molecules, implanting them on the surface to mix with the iron. At the right times, that is, when the Moon is protected from the solar wind and oxygen is present, a chemical reaction could occur that induces oxidation.
More data is needed to determine exactly how water interacts with rock. That data could also help explain another mystery: why smaller amounts of hematite are also forming on the opposite side of the Moon, where oxygen from Earth shouldn’t be able to reach it.
More science to come
Fraeman said this model can also explain the hematite found in other airless bodies such as asteroids. “It could be that small pieces of water and the impact of dust particles are allowing the iron in these bodies to rust,” he said.
Li noted that it is an exciting time for lunar science. Almost 50 years since the last Apollo landing, the Moon is once again an important destination. NASA plans to send dozens of new instruments and technology experiments to study the Moon starting next year, followed by human missions starting in 2024, all as part of the Artemis program.
JPL is also building a new version of M3 for an orbiter called the Lunar Trailblazer. One of its instruments, the high-resolution lunar volatiles and minerals mapper (HVM3), will be mapping the water ice into permanently shaded craters on the Moon, and may also be able to reveal new details about the hematite.
“I think these results indicate that there are more complex chemical processes that occur in our solar system than has been previously recognized,” Sun said. “We can better understand them by sending future missions to the Moon to test these hypotheses.”
[ad_2]