[ad_1]
Researchers in the United States say that the variant of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) – P.1 – that has emerged recently in Brazil shows a worrying level of resistance to neutralization by antibodies.
The SARS-CoV-2 virus is the agent responsible for the coronavirus disease 2019 (COVID-19) pandemic that represents a constant threat to humanity and has now claimed the lives of more than 2.56 million people in all the world.
The team at Columbia University in New York found that, in addition to being resistant to various neutralizing monoclonal antibodies (mAbs), the P.1 variant is also six times more resistant to neutralization by convalescent plasma and more than twice as resistant to sera from vaccinates than wild-type virus.
However, the loss of neutralizing activity shown by convalescent plasma and vaccine sera against P.1 was not as significant as the loss of activity reported against the B.1.351 variant identified in South Africa.
David Ho and his colleagues say that this suggests that the risk of reinfection and decreased vaccine efficacy posed by P.1 may not be as severe as those posed by B.1.351.
A pre-printed version of the research paper is available at the bioRxiv* server, while the article undergoes peer review.

Recent variants show resistance to neutralization.
Several studies have shown recently identified SARS-CoV-2 variants B.1.1.7 and B.1351 in the UK and South Africa, respectively. These carrier mutations confer resistance to neutralizing activity induced by previous infection or vaccination.
The P.1 variant that has emerged in northern Brazil has been shown to contain ten mutations in the viral spike protein, the backbone that SARS-CoV-2 uses to bind and infect cells.
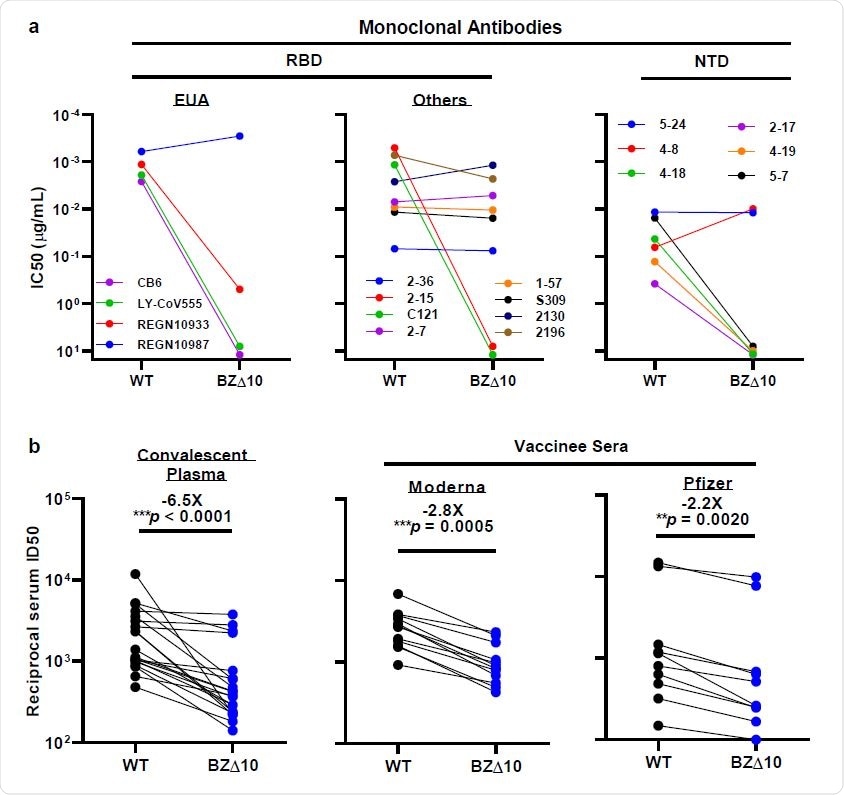
Neutralization of WT and BZ pseudovirus △ 10 by mAbs, convalescent plasma 178 and vaccinated sera. a, Changes in neutralization IC50 of selected RBD and NTD mAbs. b, Changes in convalescent plasma reciprocal plasma neutralization ID50 values and reciprocal serum ID50 values for people who received the Moderna or Pfizer vaccine. The mean times change in ID50 relative to WT is written above the p-values.
In addition to the well-established D614G mutation that became dominant early in the pandemic, P.1 contains three mutations (K417T, E484K, and N501Y) in the spike receptor-binding domain (RBD), five mutations (L18F, T20N , P26S, D138Y and R190S) in the N-terminal domain (NTD), and a mutation (H655Y) near the furin cleavage site.
The three RBD mutations are the same as those found in the RBD of B.1.351, a variant that has been shown to resist neutralization by some mAbs, convalescent plasma, and vaccine sera.
“This new variant could threaten the efficacy of current mAb or vaccine therapies because it shares mutations in the same three RBD residues with B.1.351,” Ho and the team write.
What did the researchers do?
The researchers created a SARS-CoV-2 pseudovirus containing the 10 mutations (BZ∆10) found in the P.1 variant and evaluated its susceptibility to neutralization by 18 neutralizing mAbs, 20 plasma samples from convalescent individuals, and 22 samples from vaccinated serum.
When the team tested BZ∆10 against four mAbs that had received emergency use authorization (US), including imdevimab, casirivimab, bamlanivimab, and etesevimab, the only one that maintained its original neutralizing activity was imdevimab. The neutralizing activity of the remaining three was markedly reduced or undetectable.
“Here we report that P.1 is indeed resistant to neutralization by several RBD-directed mAbs, including three with USA,” write Ho and colleagues.
mAbs targeting RBD and NTD
Next, the team tested eight mAbs targeting RBD, revealing that two previously potent mAbs did not show neutralizing activity against BZ∆10.
“Overall, these findings mimic those seen for B.1.3513, which should not be surprising, as the RBD triple mutations in P.1 and B.1.351 are largely the same,” say the researchers.
The BZ∆10 pseudovirus was also highly resistant to neutralization by four of the six NTD-targeting mAbs tested. However, two mAbs targeting the antigenic surface in NTD that have completely lost neutralizing activity against B.1.3513, remained active against BZ∆10.
“The resistance profiles are markedly different for P.1 and B.1.351, reflecting their different sets of mutations in ETDs,” the team writes.
To understand the specific mutations responsible for this neutralization pattern, the researchers tested these NTD-targeting mAbs against a panel of pseudoviruses that each contained only one of the five NTD mutations (L18F, T20N, P26S, D138Y, and R190S) that were reported. found in P .1.
As expected, the two mAbs that remained active against BZ∆10 maintained neutralizing activity against all single mutation pseudoviruses. Of the remaining four mAbs, one or more of the five mutations accounted for the loss of neutralizing activity against BZ∆10.
What about convalescent plasma and vaccinated sera?
When the researchers analyzed 20 convalescent plasma samples for neutralizing activity against BZ∆10, they observed a 6.5-fold reduction in neutralizing activity against BZ∆10, compared to wild-type pseudovirus.
Finally, serum samples from 12 recipients of the Moderna mRNA-1273 vaccine and 10 recipients of the Pfizer BNT162b2 vaccine were analyzed for neutralization against BZ∆10 and wild-type pseudovirus.
A reduction in neutralization activity against BZ∆10 was observed for each sample, but the magnitude of the loss was modest (2.8 times for Moderna; 2.2 times for Pfizer), compared to the loss against pseudovirus B.1.351 (8.6 times for Moderna; 6.5 times for Pfizer).
What do the authors suggest?
“Both convalescent plasma and vaccinated serum show a significant loss of neutralizing activity against P.1, but the decrease is not as great as that reported against B.1.351,” say the researchers. “Therefore, the threat of further reinfection or decreased vaccine protection posed by P.1 may not be as severe as B.1.351.”
The team also says that since RBD mutations are mostly the same between the two variants, the difference in their susceptibility to neutralization in sera suggests that NTD mutations can significantly affect the susceptibility of SARS-CoV-2 to antibody neutralization. .
*Important news
bioRxiv publishes preliminary scientific reports that are not peer-reviewed and therefore should not be considered conclusive, guide clinical practice / health-related behavior, or be treated as established information.