It has been 17 years since the coronavirus SARS-Kovi threatened a global epidemic. Thanks to the rapid efforts to eradicate the infection, the world’s population survived the worst.
This time we were not so lucky. The only question that makes SARS-CoV-2 more contagious than its predecessor is that we are a little closer to solving it, researchers have figured out another way to get the virus into our cells.
Researchers at the Technical University of Munich, Germany, and the University of Helsinki, Finland, conducted a study that discovered a receptor called neuropilin-1, a novel coronavirus that infects our tissues.
This particular protein is abundantly abundant on the cells in the nasal cavity, making it a piece of cake for the virus to establish a home in our body, raise a virus family and then spread to a new host.
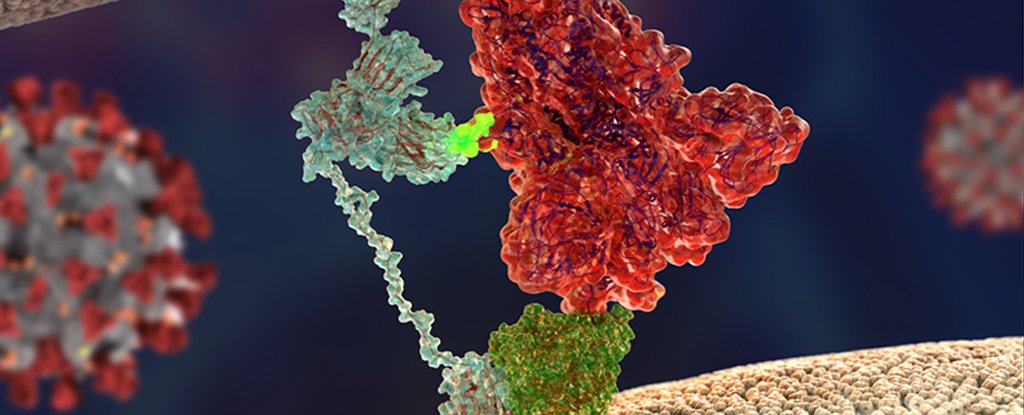
Earlier this year it was found that a receptor called angiotensin-converting enzyme 2 (ACE2) helps bind coronaviruses to the surface of cells, while an enzyme called type II transmembrane serine protease (TMPRS2) is crucial for obtaining antrim.
This type of molecular lock-picking does a good job of explaining why both SARS coronaviruses destroy so many tissues in our body, from the lining of the lungs to our digestive tract.
But that doesn’t say why one virus works better than another.
“The initial phase of our study was that SARS-CoV, a coronavirus that caused a very small disease outbreak in 2003, and SARS-CoV-2, spread differently in this way even if they use the same main receptor ACE2.” , Says University of Helsinki virologist Ravi Ojha.
A crucial part of the puzzle appeared when comparing two viral genomes; SARS-Cavi-2 minted the coin responsible for creating the prickly array of ‘hooks’, not unlike those used by other nasty pathogens to grip on host tissue.
“Compared to its old relative, the new coronavirus got an ‘extra piece’ on its surface protein, which is also found in the spikes of many destructive human viruses, including Ebola, HIV, and many pathogenic strains of Evin influenza,” said Viroilo of Helsinki University. , Says Oli Vaplahati.
“We thought this could lead us to the answer. But how?”
In consultation with colleagues around the world, the researchers zeroed in on neuropylene-1 as a common factor.
Typically, these receptors play a role in the development of tissues, especially in response to important factors between nerves. But for many viruses, it is a convenient handle to hold host cells longer to break.
Electron microscopy of the surface spikes coating SARS-Cavi-2 particles precisely indicates the possibility of a relationship with the receptor.
To help confirm this, the researchers used selected monoclonal antibodies to block the block access of different types of neuropilin-1 in the garden, but tweaked the mutations to have a slightly different composition.
Sure enough, the ‘pseudovirus’ sports SARS-Co-2 protein (excellent for watching the virus enter cells without having to worry about the business of the disruptive replication that followed) got a much harder time getting inside when Neuropylene-1 was added. Was.
“If you consider ACE2 as a door lock to enter a cell, then neuropylene-1 may be a factor that leads the virus to the door,” says Ballister.
“ACE 2 is expressed at a very low level in most cells. Thus, it is not easy to find a gateway for the virus. Other factors, such as neuropilin-1, can help the virus to find its gateway.”
With Neuropylene-1 expressed in large amounts in the nerve tissues inside the nasal cavity, we can imagine that at the moment of sniffing any infected droplets in SARS-Cavi-2 a suitable red carpet is rolled for it.
Looking at tissue samples showing neuropilin-1 taken from deceased COVID-19 patients raised suspicions, while an experiment involving mice helped confirm the role of receptors in facilitating the entry of the virus into our nervous system.
Whether this will help explain why SARS-COV-2 infection can have a traumatic effect on brain function is a question for future research.
“We can determine whether neuropilin-1, at least under the conditions of our experiments, promotes transport to the brain, but we cannot conclude whether this is also true for the stork-covi-2. “Most patients are suppressed by the immune system,” says Mika Simmons, a neurologist at the Technical University of Munich.
It’s fascinating to portray new forms of antiviral drugs on the horizon. Although SARS-CoV-2 exposes its criminal genes as quickly as possible, blocking cell receptors is bad news for our health.
That is not to say that discovery is not without opportunity.
“Currently our laboratory is testing the effect of new molecules that we specifically designed to disrupt the connection between the virus and neuropilin,” says Balisteri.
“Preliminary results are very promising and we hope to receive recognition In vivo In the near future. “
This research was published Science.
.