[ad_1]
Richter Plc. Production of a remdesivir study preparation for clinical trials began, which proved effective against coronavirus for the first time, already in the summer. By December, thousands of doses were likely to have been produced in the first round. (Richter did not want to provide the exact number to the Index.) The company is supposed to be ready for the second batch and quality control at the end of February, according to Professor Gábor L. Kóvács, director of the Hecrin Consortium, which groups together universities and large hospitals. for us, adding that it does not have accurate prognostic calculations.
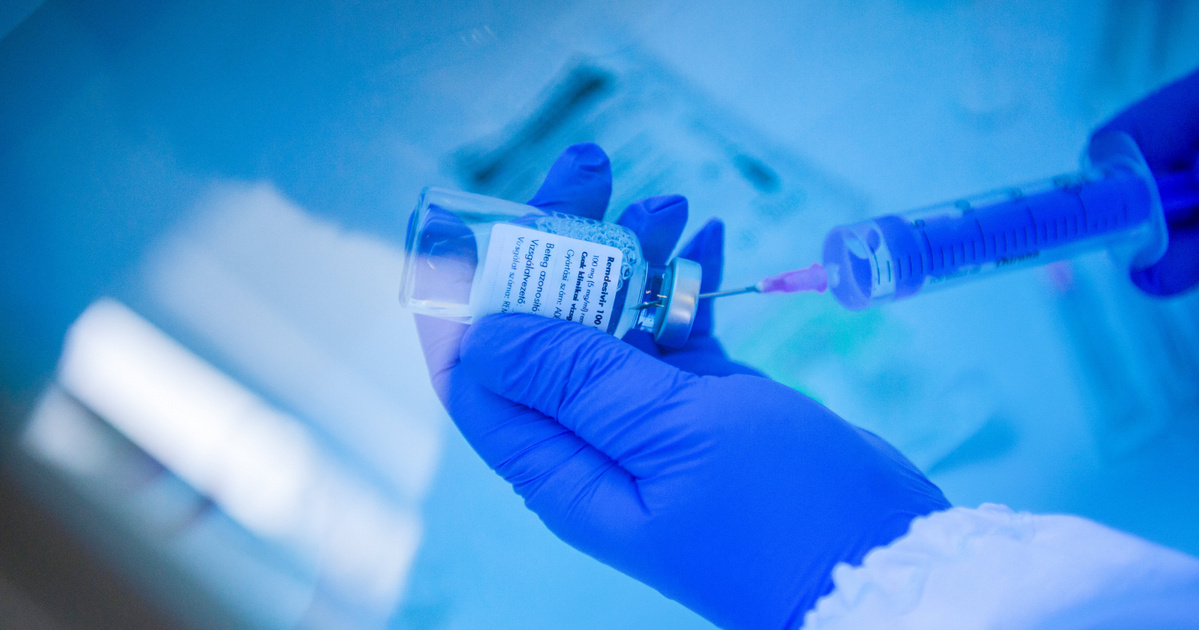
Remdesivir is an antiviral agent that is administered as an intravenous infusion to patients with moderate to severe coronavirus. In Hungary, it was initially used in clinical trials in only a few hospitals. However, for a few weeks, additional patients were discontinued in the clinical trial to obtain as many healthcare providers as possible for the drug. This was resolved by the fact that OGYÉI gave permission for pre-indication use in view of the emergency, which is a significantly less administrative procedure than a traditional clinical trial. This administration can already be carried out by smaller hospitals.
Gábor L. Kovács, head of the Hungarian Clinical Research Infrastructure Network (Hecrin), now told Index that about half of the 78 hospitals that had been delivered remdesivir under the new regulation received remdesivir in mid-December.
Therefore, a total of approximately 1,500 patients have received home remdesivir to date, 540 in the previous clinical trial, and 950-960 additional patients thereafter.
Although the Hecrin Consortium has stopped enrolling new patients in a remdesivir clinical trial, the process can be restarted at any time within a day. The Hecrin Consortium is evaluating the studies so far. The electronic recording of incoming clinical data is still incomplete by hospitals, but according to the professor, the main reason is not negligence, but the overload of doctors due to the viral situation:
The fact is, clinical trials weren’t invented for non-combat times, and lightning fast and accurate results recording. Now we need to fill in the missing data one by one for each patient. This can take weeks.
It seems likely that the first round of remdesivir won’t last until the end of February, but the new batch will only be available thereafter. The solution may be to buy new kits from the original manufacturer of remdesivir, a US company called Gilead, until the national source can provide them. This is because the US manufacturer has already closed clinical trials and approved the use of remdesivir (Veklury) by the pharmaceutical authority, so there is no need for new clinical trials, but access to the drug is currently very limited and very expensive.
Treatment with favipiravir
On Saturday we wrote about the final authorization of the other coronavirus drug, favipiravir can now be administered to patients not only with an emergency emergency approval, but also with a final authorization, based on the OGYÉI green signal.
To date, approximately 4.5 million favipiravir tablets have been purchased in Japan and China, and patients with mild symptoms of coronavirus can receive this drug. 16 tablets should be taken on the first day of therapy and six tablets from the second day. A treatment lasts 6 to 8 days, during which a patient takes an average of 70 tablets. Based on this, the current internal supply is sufficient for the therapy of around 65 thousand patients. According to our information, Egis planned to produce around 300,000 tablets in early December, and if they progressed, it would add about 4,000 more patients to treatment. Here, however, Hungarian-registered pharmaceutical companies are expected to appear.
We wrote about the latest authorized coronavirus, bamlanivimab, here!
(Cover image: Remdesivir, a drug used to treat coronavirus patients, is syringed by a protective physician in the intensive care unit for coronavirus-infected patients at St. John’s Hospital on December 9, 2020. Photo: Zoltán Balogh / MTI)
[ad_2]