[ad_1]
Dr John Raine, head of the UK Medical Regulatory Agency, reported on these reactions when he testified before a parliamentary committee on Wednesday.
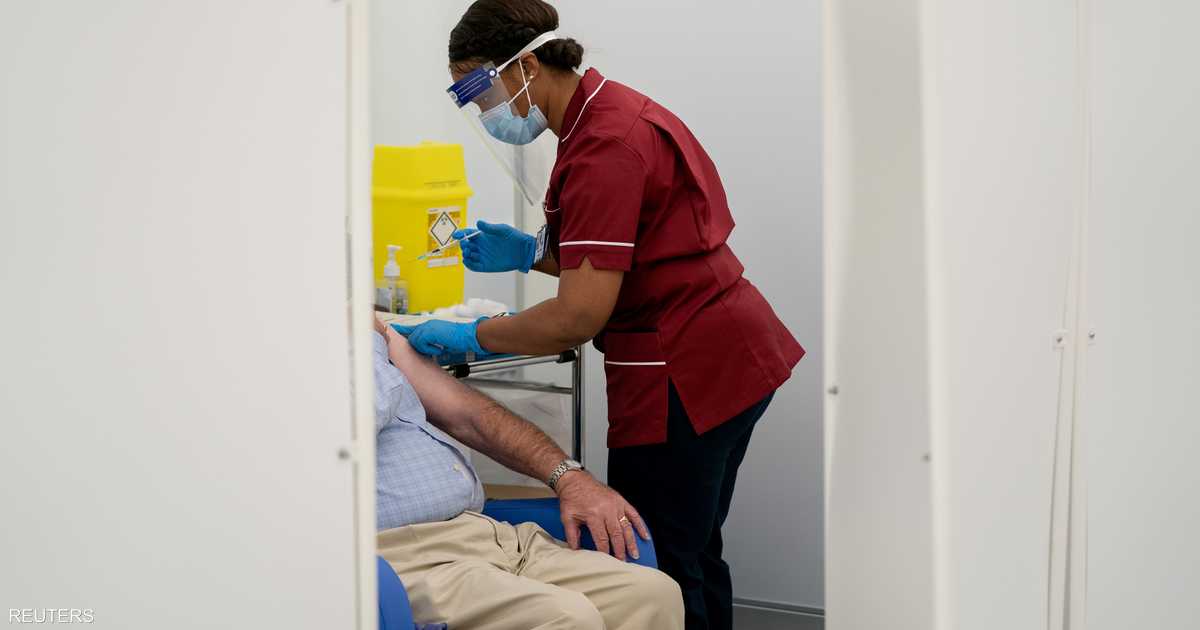
Britain began vaccinating the elderly and medical workers with the vaccine developed by the American pharmaceutical company. "Pfizer"and a company "Bountech" German, Tuesday, first vaccination in the world with this vaccine.
Raine said: "We are analyzing two cases of allergy reports. We knew from extensive clinical trials that this was not a private matter.".
And the health official continued: "But if we need to strengthen our advice, now we have this experience with vulnerable populations and groups that have been selected as a priority, we are getting that advice in the field immediately.".
Raine’s comments came as part of a public discussion about how his agency continues to monitor people who receive a vaccine. "Pfizer" Y"Bountech"Which was approved for emergency use last week.
And clinical trials had shown the vaccine’s effectiveness by more than 90 percent, knowing that the body begins to build immunity against the Corona virus days after receiving its first dose.
“>
The organizers received two reports on “possible allergic reactions” from people who participated in the first day of the comprehensive vaccination program against “Covid 19”.
Dr John Raine, head of the UK Medical Regulatory Agency, reported on these reactions when he testified before a parliamentary committee on Wednesday.
Britain began vaccinating the elderly and medical workers with the vaccine developed by the US pharmaceutical company “Pfizer” and the German company “Biontech” on Tuesday, which is the first vaccine in the world to use this vaccine.
“We are looking at two cases of allergy reports. We knew, through extensive clinical trials, that this was not a private matter,” Raine said.
And the health official added: “But if we need to strengthen our advice, now we have had this experience with vulnerable populations and groups that have been selected as a priority, and we are delivering this advice to the field immediately.”
Raine’s comments came as part of a public discussion about how his agency continues to monitor people who receive the Pfizer and Biontech vaccine, which was approved for emergency use last week.
And clinical trials had shown the vaccine’s effectiveness by more than 90 percent, knowing that the body begins to build immunity against the Corona virus days after receiving its first dose.
[ad_2]